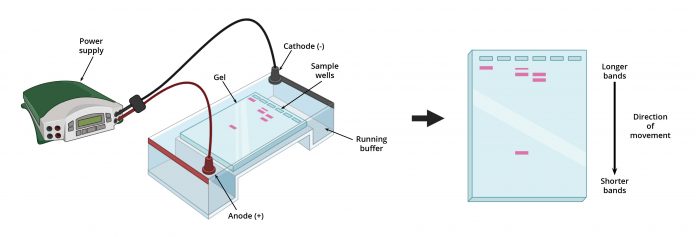
All biological macromolecules have two inescapable features, they have mass and they either have an inherent electrical charge or can be easily made to carry one. Taking advantage of these features allows for the utilization of one of the most common laboratory techniques in use today, gel electrophoresis. Using this technique, molecules of DNA, RNA or protein can be separated from one another according to their size.
What Is a Gel?
In order to separate molecules according to their size first requires a separation medium. The most commonly used media for preparing gels is either agarose, a complex polysaccharide isolated from seaweed, or polyacrylamide, a plastic polymer that can be prepared and polymerized under numerous conditions. For both agarose and polyacrylamide, solutions can be prepared and “cast” into various devices to produce a ‘slab’ with varying concentrations of the separating medium. By changing the concentrations of the medium components, the size of pores within the gel can be made to be different. Bigger molecules take longer to go through smaller pores than do small molecules and thus the first feature for fractionation of molecules by their size is established.
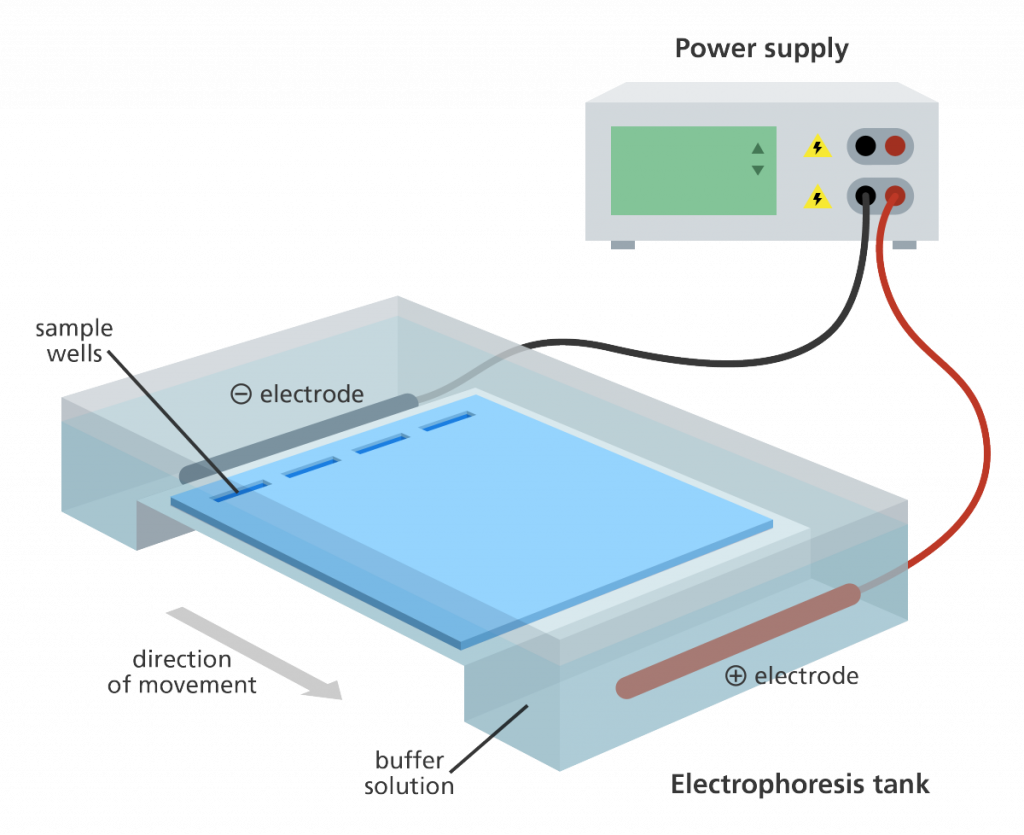
How Do the Molecules Move?
Whether the sample to be analyzed is a protein mixture or a mixture of nucleic acids, the force that makes them move through the gel is electric force. An electric field is applied across two electrodes, through a solution in which the gel is bathed and charges move between the electrodes. For any charged molecule in the gel, the electric field will force them to move, but only if they have a net electric charge.
For DNA and RNA this is an inherent property. The “backbone” of nucleic acids is made up of a chain of sugar-phosphate moieties which have a natural net negative electrical charge. Proteins do not necessarily have a net electrical charge unless they are in a solution at a pH well away from what is known as their isoelectric point (pI). However, suspending proteins in solutions containing specific detergent molecules (such as sodium dodecyl sulfate) can impart a net negative charge on almost all protein molecules and it does it in a uniform manner in solutions of various pH ranges.
If all of the molecules in a complex sample mixture can be made to have an equivalent net charge, they will all move at the same speed in a given electric field. By forcing them to go through pores of fixed sizes in either agarose or polyacrylamide, the gel matrix can affect a fractionation based on their size. Smaller molecules will get through pores more quickly. If a complex sample from, for example, a piece of pancreas is run on a polyacrylamide gel, it is now possible to separate the hormone insulin (only a few dozen amino acids long) away from the insulin receptor protein (many hundreds of amino acids long) and then detect them by means such as specific antibody staining.